
World-Class, Award-Winning RCT Proven in Real-World Clinical Settings
Validated through a multi-site, ethics-approved randomized controlled trial—supported by peer-reviewed publications and international recognition. This is a regulator-grade clinical research.
Quick Links:
At A Glance
Study Populations
1,160
Control Group:541
Intervention Group: 565
Medication Adherence (6 months)
88,4%
+24% absolute from baseline
vs. 66.7% control group
Cost Savings
-37%
IG vs. CG (excl. hospitalization)
Clinical Outcomes (IG @ 6 months from baseline)
Diabetes (HbA1c)
-1.0%
absolute IG vs. +0.2% CG
Asthma (ACQ-6 score)
-0.63
IG vs. -0.27 in CG
Heart Failure (EF%)
+50%
relative from baseline
Cholesterol (LDL mmol/L)
-0.13
IG vs. +0.28 in CG
Hypertension (SBP mmHg)
-10
IG vs. +1.3 CG
Quality of Life (AQoL-8D)
0.56 → 0.68
major improvement
The impact is clinically and statistically significant. HbA1c impact is one of the highest from any mHealth tool globally. Asthma patients on CareAide are 2x more likely to control their condition compared to standard care.
Adherence Improvements
Patient Population
1,160
Control Group
541
Intervention Group
565
Measurement Methods
PDC (Proportion of Days Covered):
Record at hospital pharmacy
MMAS-8:
Questionnaire at hospital
PDC (Proportion of Days Covered)
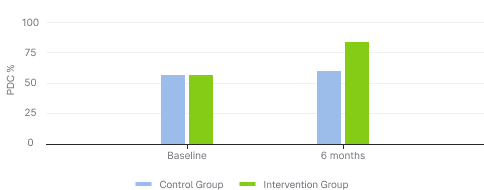
+24% absolute increase in adherence (PDC) from baseline to 6 months
MMAS-8 Score
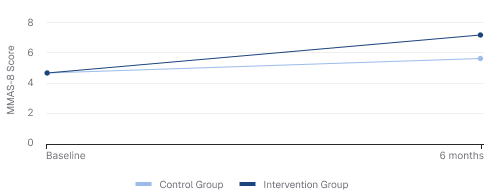
+47% improvement from baseline MMAS-8 score
Key Points

24 percent-point absolute increase in adherence (PDC)

~2X more patients achieving adherent status

Consistent improvement across income groups, including low-income B40 group

World-class results achieved by reducing treatment burden, without increasing patient effort
CareAide is not just a tracker, it is a Social Adherence Platform.
When adherence infrastructure is automated and socially reinforced, adherence becomes the default—not the exception.
Clinical Outcomes
Detailed Clinical Measurements: Baseline vs 6 Months
| Condition / Biomarker | Measurement | CG Baseline | CG 6 Months | IG Baseline | IG 6 Months | Change (IG vs CG) | Target Range* |
|---|---|---|---|---|---|---|---|
| Hypertension | SBP (mmHg) | 142.0 | 147.3 | 146.3 | 136.2 | -10.1 vs +5.3 | 120-129 High-normal |
| DBP (mmHg) | 75.5 | 78.6 | 77.1 | 71.6 | -5.5 vs +3.1 | < 80 | |
| Cholesterol | LDL (mmol/L) | 3.03 | 3.32 | 2.65 | 2.52 | -0.13 vs +0.29 | < 2.6 |
| Diabetes | HbA1c (%) | 8.4 | 8.2 | 8.5 | 7.3 | -1.0 vs -0.2 | < 7.0% |
| Asthma | ACQ-6 score | 1.45 | 1.35 | 1.47 | 1.03 | -0.44 vs -0.10 | < 0.75 = Well-controlled |
| Heart Failure | NYHA Class | 1.86 | 1.92 | 1.97 | 1.63 | -0.34 vs +0.02 | Class I = No limitation |
*ESC/ESH 2018; MOH Malaysia HTN CPG 2023; AHA/ACC 2018 Lipid Guideline; ADA 2024; WHO 2020 Diagnostic Criteria; GINA 2023; ACC/AHA 2022 HF Guideline.
Table Notes: CG = Control Group, IG = Intervention Group. All measurements taken at baseline and 6-month follow-up. Lower scores indicate better outcomes for all metrics except NYHA class where lower is better.
Change from Baseline Comparison
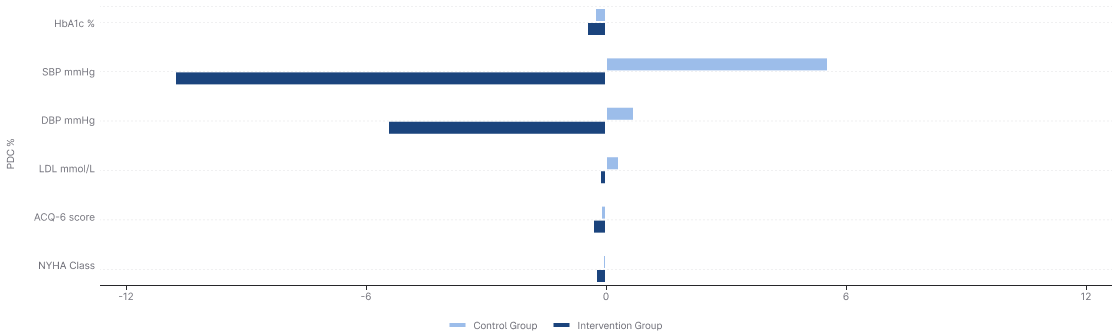
Statistical Significance
All primary endpoints achieved statistical significance (p < 0.05) with clinically meaningful effect sizes.
Global Benchmark
The HbA1c reduction of -1.0% absolute is among the highest documented from any mHealth intervention globally.
Healthcare Cost Savings
Direct Medication Saving
RM 360K
Per 1,000 patients annually through optimized medication use and better adherence
Medication Adherence (6 months)
RM 3M+
Per 1,000 patients annually by preventing disease progression and complications
IG consume less medicine (avg. per month savings)
Extrapolate to 1K patients/year
RM 360K/year
Cost Savings Comparison (RM/month)

| Disease | IG Cost (RM) | CG Cost (RM) | Δ Cost (RM) | PDC Impact |
|---|---|---|---|---|
| Hypertension | 220 | 250 | -30 | 98% vs. 85% |
| Diabetes | 310 | 340 | -30 | 100% vs. 67% |
| Asthma | 180 | 210 | -30 | 95% vs. 50% |
| Heart Failure | 400 | 420 | -20 | 93% vs. 83% |

Important note on cost interpretation
Cost savings reported in this study reflect the Malaysian public health system, where many services are heavily subsidised or free at the point of care. Actual savings for your system may vary depending on funding model, tariffs, and patient mix.
Patient Quality of Life – Did They Feel Better themselves?
Patients Felt Healthier and More in Control
The CareAide app meaningfully enhanced patients' overall well-being and day-to-day functioning.
Changes in Quality of Life Over Time (AQoL-6D Scores)
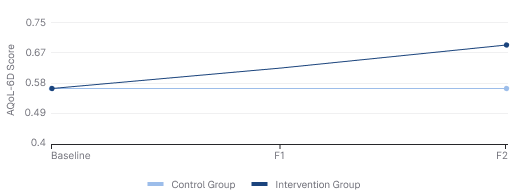
0.56 → 0.68
Intervention Group Improvement
EQ-5D Mean Score Over Time

2.22 → 2.08
Intervention Group Improvement (lower is better)
Productivity Gains (All Diseases)
A turnkey RPM solution that strengthens health outcomes and patient loyalty—without expanding your clinical team.
RM 5.24M
Total annual net productivity gain per 1K workforce
| Component | IG Savings (per patient/year) | CG Losses (per patient/year) | Net Benefit (per patient/year) |
|---|---|---|---|
| Absenteeism (average days/patient/year) | RM 840 (5.6 days) | RM 2,115 (14.1 days) | +RM 1,275 (8.5 days) |
| Productivity score: 6.1/10 (±1.2) → 27% higher vs. 4.8/10 (±1.5) | 27% of 240 working days/year = 65 regained productive days. 65 × RM60 = +RM 3,900 | ||
| Total (Workforce Only) | +RM 5,175 |
Absenteeism Reduction
Presenteeism Improvement
Methodology Notes:
- Self-reported productivity scale (0=unproductive, 10=fully productive)
- Absenteeism uses RM 150/day based on the reported data
- Presenteeism is valued at 40% of wage (RM 60/day) to reflect partial productivity losses
About the Research (RCT Overview)
Our Study at a Glance: Key Facts of the RCT CareAide®
Pioneering study, one of the largest/deepest in the region, cultivating robust, homegrown evidence to illuminate pathways to a healthier, more vibrant Malaysia.
Design
A 6-month, prospective Randomized Controlled Trial (RCT).
Scale
1,106 patients across 3 major Malaysian hospitals (UMMC, H. Putrajaya, H. Pulau Pinang).
Participants
Adults with Hypertension, Diabetes, Asthma, or Heart Failure who had low baseline medication adherence (MMAS Score less than 6).
Method
IG: Received standard care + used the CareAide® App. CG: Received standard care only.
Total Participants
1,106
Intervention Group
565
Control Group
541
Peer-Reviewed Publications
Primary RCT Outcomes Publication
Published in peer-reviewed medical journal
Comprehensive report of the multi-site randomized controlled trial demonstrating the efficacy of the CareAide platform in improving medication adherence and clinical outcomes across multiple chronic conditions.

Scientific Posters & Presentations
Presented at international medical conferences
Research findings presented at major international conferences including findings on cost-effectiveness, quality of life improvements, and condition-specific outcomes.

Cost-Effectiveness Study
Health-economic evaluation
Detailed health economic analysis demonstrating the cost savings and return on investment from implementation of the CareAide platform in chronic disease management.

Study Protocol & Design
Presented at international medical conferences
Research findings presented at major international conferences including findings on cost-effectiveness, quality of life improvements, and condition-specific outcomes.
Regulatory & Global Registries
Our research is registered with international clinical trial registries and recognized by global health organizations, ensuring transparency, accountability, and adherence to the highest research standards.


WHO Digital Health
Included in the World Health Organization's Digital Health Atlas (DHA), a global technology registry for digital health implementations.


Clinical Trial GOV
Registered with the U.S. National Library of Medicine's clinical trial registry, the world's largest database of privately and publicly funded clinical studies.


NMRR Malaysia
Registered with the National Medical Research Register, Malaysia's official repository for medical and health research conducted in the country.

Research Integrity & Transparency
Registration with these international bodies demonstrates our commitment to research transparency, ethical conduct, and compliance with international standards for clinical research. All outcomes were pre-specified and the study followed Good Clinical Practice (GCP) guidelines.

Ready to Achieve Similar Outcomes?
Join healthcare organizations worldwide leveraging evidence-based RPM.